whats happening to dcpip sitting in the tube prior to the experiment starting
It is fairly easy to show that plants produce oxygen and starch in photosynthesis. At historic period xiv–16 students may have collected the gas given off by pond weed (for example Elodea) and tested leaves for starch.
Information technology is not quite so easy to demonstrate the other reactions in photosynthesis. For the reduction of carbon dioxide to carbohydrate at that place must exist a source of electrons. In the jail cell, NADP is the electron acceptor which is reduced in the lite-dependent reactions, and which provides electrons and hydrogen for the light-independent reactions.
In this investigation, DCPIP (two,six-dichlorophenol-indophenol), a blue dye, acts equally an electron acceptor and becomes colourless when reduced, allowing whatsoever reducing agent produced past the chloroplasts to be detected.
Lesson organisation
This investigation depends on working quickly and keeping everything cool. Your students will demand to empathise all the instructions in advance to exist certain that they know what they are doing.
Apparatus and Chemicals
Per student or grouping of students:
Centrifuge – with RCF between 1500 and 1800g
Centrifuge tubes
Fresh green spinach, lettuce or cabbage, 3 leaves (discard the midribs)
Scissors
Common cold pestle and mortar (or blender or food mixer) which has been kept in a freezer compartment for 15–30 minutes (if left too long the extract will freeze)
Muslin or fine nylon mesh
Filter funnel
Ice-water-salt bath
Glass rod or Pasteur pipette
Measuring cylinder, 20 cm3
Chalice, 100 cm3
Pipettes, 5 cm3 and i cm3
Bench lamp with 100 West bulb
Examination tubes, 5
Humid tube
Pipette for v cm3
Pipette for 0.v cm3
Pipette filler
Waterproof pen to label tubes
Colorimeter and tubes or calorie-free sensor and information logger
0.05 M phosphate buffer solution, pH 7.0: Shop in a refrigerator at 0–4 °C (Note 1).
Isolation medium (sucrose and KCl in phosphate buffer): Store in a refrigerator at 0–4 °C (Notation two).
Potassium chloride (Low Chance) (Note 3).
DCPIP solution (Depression Gamble): (i x 10- 4 G approx.) (Note 4)
Health & Safety and Technical notes
Although DCPIP presents minimal hazard apart from staining, information technology is best to avoid peel contact in case prolonged contact with the dye causes sensitisation.
Do not handle electric light bulbs with wet hands.
All solutions used are depression take a chance – refer to relevant CLEAPSS Hazcards and Recipe cards for more information.
ane 0.05 Thousand phosphate buffer solution, pH 7.0. NatwoHPO4.12H2O, 4.48 g (0.025 M) KH2PO4, 1.seventy g (0.025 M). Make upward to 500 cmthree with distilled water and store in a fridge at 0–4 °C. Depression run a risk – refer to CLEAPSS Hazcard 72.
2 Isolation medium. Sucrose 34.23 g (0.4 M) KCl 0.19 g (0.01 M). Dissolve in phosphate buffer solution (pH 7.0) at room temperature and brand up to 250 cm3 with the buffer solution. Store in a fridge at 0–iv °C. Depression hazard – refer to CLEAPSS Hazcard 40C.
3 Potassium chloride 0.05 M. Deliquesce 0.93 1000 in phosphate buffer solution at room temperature and make upwardly to 250 cmiii. Store in a refrigerator at 0–four °C. Use at room temperature.(Note that Potassium chloride is a cofactor for the Hill reaction.) Refer to CLEAPSS Hazcard 47B and Recipe card 51.
4 DCPIP solution DCPIP 0.007–0.01 g, fabricated upward to 100 cm3 with phosphate buffer. Refer to CLEAPSS Hazcard 32 and Recipe menu 46.
Procedure
Keep solutions and apparatus cold during the extraction process, steps 1–eight, to preserve enzyme activity. Acquit out the extraction every bit quickly as possible.
Preparation
a Cut three small green spinach, lettuce or cabbage leaves into small pieces with pair of scissors, simply discard the tough midribs and leaf stalks. Place in a cold mortar or blender containing 20 cm3 of cold isolation medium. (Scale up quantities for blender if necessary.)
b Grind vigorously and rapidly (or blend for about 10 seconds).
c Place four layers of muslin or nylon in a funnel and wet with cold isolation medium.
d Filter the mixture through the funnel into the beaker and cascade the filtrate into pre-cooled centrifuge tubes supported in an water ice-water-table salt bathroom. Gather the edges of the muslin, wring thoroughly into the chalice, and add filtrate to the centrifuge tubes.
east Check that each centrifuge tube contains about the same volume of filtrate.
f Centrifuge the tubes for sufficient time to get a minor pellet of chloroplasts. (10 minutes at high speed should be sufficient.)
chiliad Pour off the liquid (supernatant) into a boiling tube existence conscientious non to lose the pellet. Re-suspend the pellet with about 2 cmthree of isolation medium, using a glass rod. Squirting in and out of a Pasteur pipette five or six times gives a compatible suspension.
h Store this foliage extract in an ice-water-salt bath and employ as soon as possible.
Investigation using the chloroplasts
Read all the instructions earlier you start. Use the DCPIP solution at room temperature.
i Set up 5 labelled tubes equally follows.
| Tube | Leafage extract (cm3) | Supernatant (cm3) | Isolation medium (cm3) | Distilled water (cm3) | DCPIP solution (cm3) |
| 1 | 0.5 | – | – | – | 5 |
| 2 | – | – | 0.5 | – | v |
| iii | 0.v | – | – | – | 5 |
| 4 | 0.5 | – | – | 5 | – |
| five | – | 0.5 | – | – | 5 |
j When the DCPIP is added to the extract, milkshake the tube and note the fourth dimension. Identify tubes ane, 2 and 4 near 12–xv cm from a vivid light (100 W). Place tube 3 in darkness.
k Time how long it takes to decolourise the DCPIP in each tube. If the extract is so agile that it decolourises inside seconds of mixing, dilute it 1:five with isolation medium and try again.
Teaching notes
Traditionally the production of oxygen and starch are used as show for photosynthesis. The light-dependent reactions produce a reducing agent. This unremarkably reduces NADP, but in this experiment the electrons are accepted by the bluish dye DCPIP. Reduced DCPIP is colourless. The loss of colour in the DCPIP is due to reducing amanuensis produced by light-dependent reactions in the extracted chloroplasts.
Students must develop a clear understanding of the link betwixt the light-dependent and lite-contained reactions to be able to interpret the results. Robert Hill originally completed this investigation in 1938; he concluded that h2o had been split up into hydrogen and oxygen. This is at present known as the Hill reaction.
You can examine a driblet of the sediment excerpt with a microscope under high power to see chloroplasts. There volition be fewer chloroplasts in the supernatant – which decolourises the DCPIP more slowly, reinforcing the idea that the reduction is the result of chloroplast activeness.
Sample results
Using a bench centrifuge
The experimental procedure was followed. A standard lab centrifuge was used to spin down the chloroplasts (Clifton NE 010GT/I) at 2650 RPM, 95 X g for 10 minutes.
The experiment was started within v minutes of preparing the chloroplasts. The reaction was followed using an EEL colorimeter with a scarlet filter – readings taken every minute.
| Time/min | Absorption Tube one | Absorption Tube five |
| ii | 5.0 | 5.0 |
| 3 | iv.6 | 4.6 |
| 4 | 4.3 | iv.0 |
| v | four.0 | 3.vii |
| 6 | 3.8 | three.four |
| seven | 3.4 | iii.0 |
| viii | 3.0 | ii.7 |
| ix | 2.half dozen | 2.6 |
| x | two.ii | 2.three |
| 11 | 1.9 | 2.0 |
| 12 | 1.4 | 1.7 |
| 13 | 0.9 | one.6 |
| 14 | 0.vi | 1.3 |
| 15 | 0.5 | 1.0 |
| sixteen | 0.v | 0.8 |
| 17 | 0.4 | 0.5 |
| 18 | 0.five | 0.three |
| 19 | 0.4 | 0.3 |
| 20 | 0.3 | 0.three |
Tube iii (incubated in the dark) gave a reading of 5.4 absorption units later on 20 minutes.
Tube 2 (DCPIP with no leaf extract) was half-dozen.2 absorption units.
Using a micro-centrifuge
The experiment was repeated using a micro-centrifuge.
| Time/min | Absorption Tube 1 | Absorption Tube 5 |
| i | 3.eight | three.v |
| 2 | 2.ix | iii.1 |
| three | 2.3 | 2.8 |
| 4 | ane.8 | 2.5 |
| 5 | ane.iii | 2.3 |
| 6 | 0.9 | ii.1 |
| 7 | 0.7 | 1.8 |
| viii | 0.6 | 1.6 |
| 9 | 0.6 | i.three |
| 10 | 0.6 | 1.1 |
Tube iii (incubated in the nighttime) gave a reading of 4.ix assimilation units afterwards 10 minutes.
Tube 2 (DCPIP with no leaf extract) was six.four absorption Units.
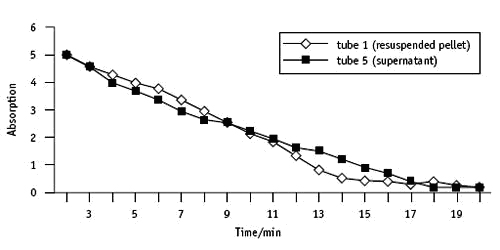
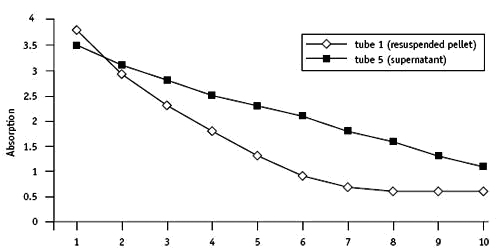
The relative activeness of the pellet was higher than when the bench centrifuge was used. The micro-centrifuge tubes were merely 1.five cmthree capacity – not ideal for this practical. A higher speed bench centrifuge would be better.
In club to check for loss of chloroplast activity, the experiment was repeated using the same chloroplast suspension 1 and two hours after preparation. Chloroplast suspension was kept in a salt-water ice bath. There was no loss of activity when the extract was kept in ice for upward to 2 hours.
Pupil questions
1 Depict and explicate the changes observed in the five tubes. Compare the results and make some last comments most what they show.
ii The charge per unit of photosynthesis in intact leaves can be limited by several factors including low-cal, temperature and carbon dioxide. Which of these factors will have little effect on the reducing capacity of the leafage extract?
3 Describe how y'all might extend this applied to investigate the event of low-cal intensity on the light-dependent reactions of photosynthesis.
Answers
1 Colour change and inferences that tin fabricated from the results:
Tube one (leaf extract + DCPIP) colour changes until it is the same colour equally tube 4 (leaf excerpt + distilled water).
Tube 2 (isolation medium + DCPIP) no colour alter. This shows that the DCPIP does not decolourise when exposed to light.
Tube 3 (leafage extract + DCPIP in the night) no color change. Information technology can therefore be inferred that the loss of color in tube i is due to the effect of low-cal on the extract.
Tube 4 (leaf extract + distilled water) no colour change. This shows that the extract does not change colour in the light. It acts every bit a colour standard for the extract without DCPIP.
Tube 5 (supernatant + DCPIP) no colour change if the supernatant is clear; if it is slightly green there may exist some decolouring.
The results should indicate that the light-dependent reactions of photosynthesis are restricted to the chloroplasts that have been extracted.
2 Carbon dioxide will have no effect, because it is non involved in the light-dependent reactions.
3 Students should describe a procedure in which low-cal intensity is varied but temperature is controlled.
Health and rubber checked, September 2008
Related experiment
Investigating photosynthesis using immobilised algae
Source: https://practicalbiology.org/energy/photosynthesis/investigating-the-light-dependent-reaction-in-photosynthesis
0 Response to "whats happening to dcpip sitting in the tube prior to the experiment starting"
Post a Comment